Global Trials, Local Limits
How geography, infrastructure, and everyday logistics shape who can participate
The appointment is calm in the way medical appointments often are. The diagnosis is explained carefully, the treatment plan outlined step by step. There are follow-up labs, a medication to begin next week, another visit already placed on the calendar before she leaves the room. Nothing feels chaotic. It feels deliberate. Structured. Designed to move forward.
That structure is oddly comforting. It suggests that someone, somewhere, knows what comes next.
Later that evening, when the house is quiet and the day has settled, she opens her laptop and searches anyway. Not because she doubts her doctor, and not because she wants to abandon the plan already in motion. But because once a condition has a name, it also has research attached to it. Somewhere, someone is studying it. Somewhere, something new is being tested.
The website she finds is dense and difficult to navigate. The language reads as if it were written for regulators and sponsors rather than patients trying to understand their options. Inclusion criteria stretch on for paragraphs. Acronyms stack up. It takes effort to determine whether any of it applies to her.
One study catches her attention. It is described as multinational. Multi-site. Global. The word feels expansive. Serious. As if the study stretches far beyond the walls of the clinic she visited that morning.
She scrolls down to the list of locations.
They are precise. Cities in other states. Academic centers she has heard of but never visited. One site across a border. Another overseas.
The trial exists. It is active. It is enrolling.
It is not here.
There is an email address listed. She writes. There is a phone number. It routes to voicemail. The outgoing message is generic, professional, unconnected to the name of the study she just read about.
At her next appointment, she brings it up. Her physician hasn’t heard of the trial. He opens the same registry page and scans it quickly. He says he can try to find out more. The conversation returns to the treatment already underway.
Nothing in this sequence feels dramatic. No one says no. No one denies her access.
And yet something has shifted. The trial, which moments before felt like possibility, now feels theoretical — a study happening somewhere else, for someone else, under conditions that do not quite extend to her life.
This is where the idea of a “global” clinical trial begins to narrow. Not in the protocol. Not in the consent form. But in the quiet realization that existence is not the same thing as reach.
Where Trials Actually Take Place
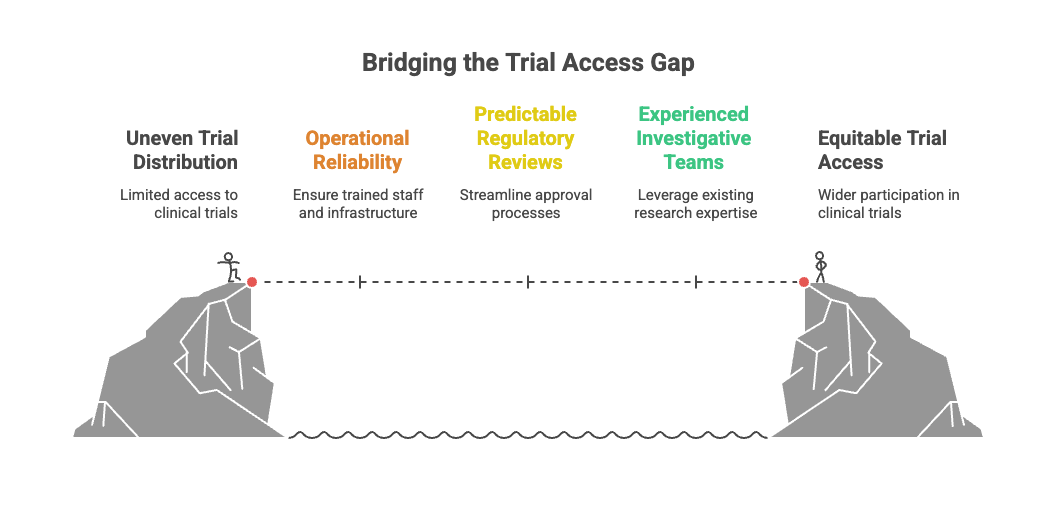
Clinical trials do not distribute themselves evenly across a map. They cluster.
They cluster around large academic medical centers, established research hospitals, and regions with experienced investigative teams. They return to cities where enrollment targets were previously met, where regulatory reviews move predictably, where coordinators know the rhythm of a protocol and sponsors know what to expect.
From an operational perspective, this makes sense. Research requires reliability. It requires trained staff, calibrated equipment, pharmacy controls, safety reporting systems, and oversight mechanisms that meet regulatory standards. It requires infrastructure that can sustain not just enrollment, but follow-up, data capture, and adverse event management.
A “global” trial often means that multiple countries are represented. It does not mean that participation is evenly accessible within those countries.
For the woman at her kitchen table, this distinction is not theoretical. It determines whether the trial is something she can step into or something she can only read about.
What It Takes for a Site to Host a Trial
Before any patient can consider participation, a site must be capable of hosting the study.
That capability depends on more than clinical expertise. It depends on regulatory bodies that can review protocols in a reasonable timeframe. Ethics committees that can monitor ongoing conduct. Laboratories that meet certification standards. Imaging equipment that aligns with protocol specifications. Supply chains that can transport investigational products across borders without disruption.
In many parts of the world, those systems exist. In others, they are developing, uneven, or burdened by resource constraints. Even within highly developed healthcare systems, research capacity is concentrated. Community hospitals may deliver excellent care but lack the staffing or certification required to conduct complex clinical trials.
Participation, then, is shaped long before an individual makes a decision. It is shaped by whether a location has the stability, oversight, and infrastructure necessary to hold the weight of a study. When research capacity clusters, opportunity clusters with it.
The protocol may be written broadly. The map is not.
Distance Is a Barrier, Even When No One Names It
When a trial is technically open to someone but practically distant, the burden shifts quietly to the individual.
Travel becomes part of the calculation. Time away from work. Childcare arrangements. Transportation costs. Border crossings. Language differences. The possibility of follow-up visits extending months or years into the future.
These are not exclusion criteria. They do not appear in bold on a registry page.
But they narrow participation all the same.
For some patients, traveling several hours for specialized care is already part of life. For others, it is not feasible. For some families, flexible work arrangements make repeated visits possible. For others, a single missed shift carries financial consequence.
Willingness is only one variable in participation. Stability is another. Proximity is another still.
When trials are described as global, these differences rarely make it into the headline.
Why Certain Regions Appear Again and Again
If you scan multinational studies over time, patterns emerge. The same countries. The same metropolitan hubs. The same high-performing sites.
Sponsors and CROs return to places where they can enroll efficiently and collect reliable data. Speed matters. Predictability matters. Prior relationships matter.
Over time, those decisions shape the geography of evidence.
Evidence follows infrastructure.
None of this is inherently wrong. Clinical development operates under timelines, funding constraints, and regulatory expectations. Choosing experienced sites reduces risk.
But over time, these decisions create an uneven geography of evidence. Research follows infrastructure. Infrastructure follows investment. Investment follows demonstrated performance.
The cycle reinforces itself.
Meanwhile, patients outside those established networks may encounter trials only as distant listings on a registry page — visible but out of reach.
Before We Talk About Results
When results are eventually published, they are often described as coming from a global study. That label carries weight. It implies breadth. Representation. Applicability.
What it does not always reveal is how participation was geographically distributed within that global frame.
If access is clustered, participation is clustered. And if participation is clustered, the lived realities represented in the data may also cluster — even if unintentionally.
Understanding this does not diminish the value of research. It clarifies its context.
The woman who searched that night may never enroll in that trial. She may continue with the treatment plan already underway. The study will proceed, data will be collected, results will be analyzed.
But the question remains: who had the practical ability to say yes?
What Global Could Mean
The woman who searched that night may never hear back from the study team. She may continue with the treatment already in motion. Months later, she may see a headline announcing that the global trial has completed enrollment. Eventually, she may read about its results.
From the outside, it will look comprehensive. International. Carefully conducted.
And it will be.
But it will also reflect the map of where research could realistically happen.
Global trials are not failures of intent. They are reflections of infrastructure. Of investment. Of where research capacity has been built and sustained over time.
The question, then, is not whether global trials are legitimate.
The question is what we want “global” to mean.
Does it mean multiple countries represented?
Or does it mean research capacity that is actively expanding into new regions?
Does it mean centralized excellence?
Or distributed access?
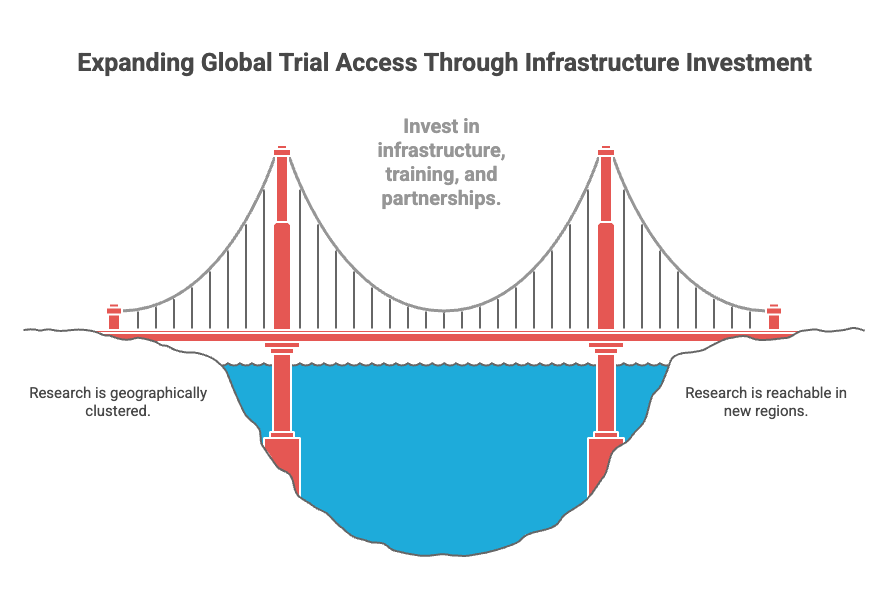
Broadening access is not as simple as adding more dots to a map. It requires long-term investment in research infrastructure, regulatory support, training, and local partnerships. It requires designing trials that can travel — operationally, culturally, and logistically — across different systems.
That work is slower. It is less visible than a press release. It does not fit neatly into enrollment timelines.
But it determines whether the next patient who searches at her kitchen table sees something that is not just global in scope — but reachable in practice.
🧭 Compass Insight
Clinical trial access is often framed as a question of willingness. In reality, it is equally a question of geography, infrastructure, and everyday stability.
“Global” describes the scope of a study. It does not guarantee its reach.
Seeing that difference clearly does not weaken trust in research. It strengthens our ability to interpret it and to design future trials that extend beyond the places where research has historically clustered.





A must read, Thanks!!!!