The Readout that Decides Everything
What Trial Results Actually Mean for Patients, Programs, and the People Behind Them
On the morning a critical trial reads out, two press releases sit open on a laptop.
Both are drafted. Reviewed. Approved.
Ready to drop into a slide deck.
Ready to publish.
One announces success.
One explains failure.
The team waits for the readout…
For patients, the result may determine whether this therapy ever reaches them.
If the data are positive, the program advances. Toward approval, partnership, acquisition, scale.
If the data fall short, hard decisions follow. Pause. Pivot. Restructure. Stop.
For the team who built the program, it may decide whether the work continues. For the company, it can redraw the future.
The data are about to be known.
When they are revealed, they determine what happens next.
What Is Actually Revealed on Readout Day
The decisions that shape a readout are made before the first patient is ever enrolled.
The protocol defined the population.
The endpoints were negotiated.
The comparator was selected.
The geography shaped who enrolled.
The statistical plan was locked.
Readout day does not introduce new science. It shows how those earlier decisions performed.
And what gets revealed is rarely simple.
The primary endpoint may be met, but narrowly. The effect size may be statistically significant but clinically modest. Safety may look acceptable overall but uneven in certain groups. A secondary endpoint may show strength even if the primary falls short.
The same dataset can look transformative in an unmet need and incremental in a crowded market.
Regulators, investors, clinicians, and competitors will interpret the data through different lenses.
Headlines compress this into a single word. The dataset does not.
A readout is not random. It reflects design choices made years earlier in protocol development, endpoint selection, geography, and statistical planning. It is the moment when that structure is tested.
When the Signal Is Positive
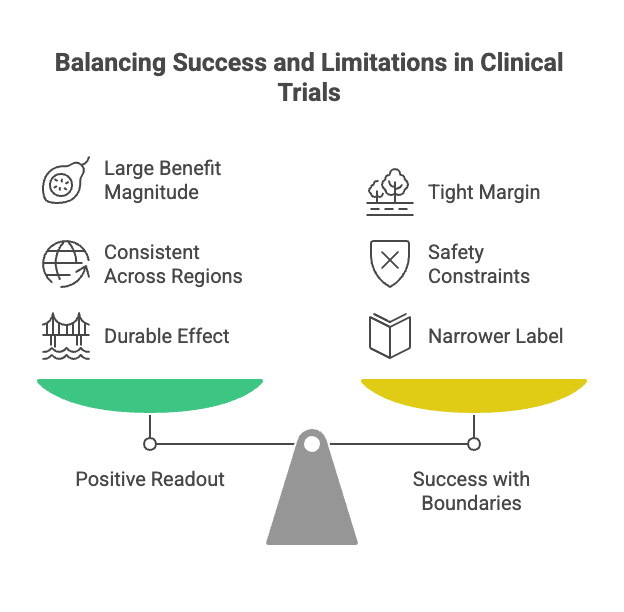
A positive readout is not one thing. It is a profile.
How large is the benefit?
Is it consistent across regions and sites?
Does it hold across age groups, disease severity, prior treatments?
Does safety meaningfully limit who can use it?
Is the effect durable?
Sometimes the separation between drug and comparator is obvious. Those moments change practice.
More often, the win has edges. The endpoint is met, but the margin is tight. Safety introduces constraints. The eventual label may be narrower than the development team expected.
Regulators will ask whether the endpoint reflects meaningful clinical benefit. Payers will ask whether the magnitude justifies cost. Competitors will measure it against their own data.
A positive readout can open regulatory pathways, accelerate partnerships, or spark acquisition interest.
It can also define the limits of the program.
Success still has boundaries.
When the Signal Falls Short
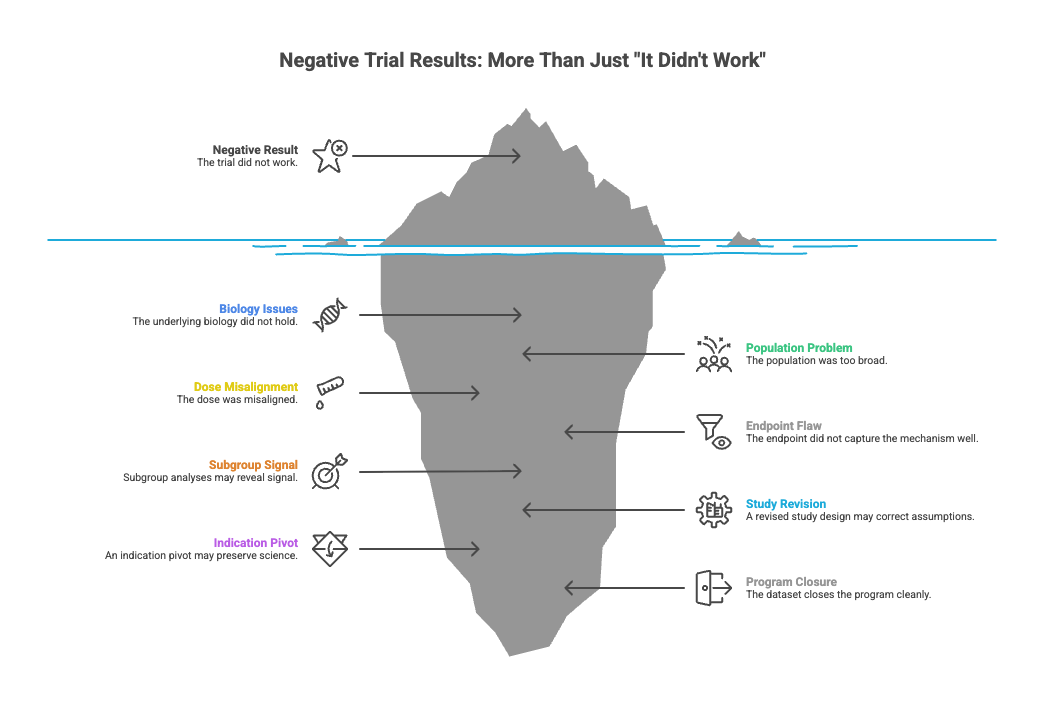
A negative result is rarely just “it didn’t work.”
Sometimes the biology does not hold.
Sometimes the population was too broad.
Sometimes the dose was misaligned.
Sometimes the endpoint did not capture the mechanism well.
Subgroup analyses may reveal signal in a narrower population. A revised study design may correct earlier assumptions. An indication pivot may preserve part of the science.
And sometimes the dataset closes the program cleanly.
That clarity matters.
A decisive failure can prevent years of incremental spending chasing marginal benefit. It can redirect capital and attention toward stronger assets. It can end uncertainty faster than cautious optimism ever could.
Negative data can still create value, but often through discipline.
The Decisions That Follow
A readout concentrates consequence.
Inside companies, the hours after a readout are quiet and intense. Teams parse tables. Subgroup analyses are reviewed again. Slide decks are updated. Regulatory briefing documents are drafted. Calls are scheduled before the public narrative settles.
Interpretation begins immediately.
For Patients
For patients, the readout changes direction.
If the data are strong, the therapy moves closer to approval. Regulatory submissions begin. Access timelines become concrete. Expanded access programs may open.
If the data fall short, development may stop. The therapy leaves the near-term landscape. Patients return to existing standards of care.
In rare diseases, entire communities may be tracking the trial. In common diseases, the impact is less visible but still real. A modest benefit may limit eligibility. A safety issue may narrow who can receive it.
Patients do not experience statistical thresholds. They experience availability, eligibility, and time.
For the Program
For the development team, the dataset becomes instruction.
A strong result accelerates planning. Regulatory meetings are scheduled. Manufacturing timelines tighten. The next trial begins to take shape.
A narrow result demands scrutiny. Additional data may be required before filing. Study design may need adjustment.
A miss forces harder questions. Continue? Refocus? Or stop?
At this level, the data do not celebrate or mourn. They guide.
For the Company
At the company level, the impact is structural.
Capital markets respond.
Partnership discussions shift. Acquisition interest can emerge after a strong Phase II signal.
A pivotal miss can reset valuation just as quickly.
Pipeline priorities move. Hiring plans change. Financing terms are renegotiated. The data narrow the range of viable futures.
What leadership does next determines whether the science moves forward.
What We Mean When We Say “Results”
When we say “the results are in,” it sounds final.
In reality, a readout answers a series of defined scientific questions about efficacy and safety within the framework that was built years earlier.
But the data do more than resolve statistical uncertainty.
They reshape access.
They redirect programs.
They alter companies.
For patients, they determine whether a therapy continues toward approval or disappears from development.
For a program, they define the next study or the last.
For a company, they influence capital, partnerships, and survival.
A trial readout does not close a chapter. It defines which future is now possible.
What we build from that data determines whether the science becomes care.





Reading this reminds me how quiet and heavy those moments must feel. So many years of belief, work, and hope distilled into a single line of data.
Behind every “result” there are people who dared to try to change what patients live with, and that kind of courage deserves to be seen, no matter what the readout says.